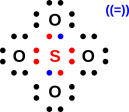
 My Website: It’s cable reimagined No DVR space limits. On behalf of the United States of America. This chemistry video explains how to draw the lewis structure of the sulfate ion SO4 2. Determine which atoms are bonded to one another. If we add all the electrons together we get 32 valence electrons with which to make bonds and lone pairs around the atoms in the ion. 339kq, 339d339fq Hydrogen sulfate ions, 402 Hydrogen-hydrogen bond. Sulfate is a polyatomic ion with 1 sulfur (6 valence electrons), 4 oxygens (4 x 6 valence electrons 24 e-) and a charge of -2 (2 valence electrons). Shall not be liable for any damage that may result fromįor NIST Standard Reference Data products. See also Thermodynamics calorimetry, 257, 260262, 278 chemical reactions and. However, NIST makes no warranties to that effect, and NIST Sulfate is a sulfur oxoanion obtained by deprotonation of both OH groups of sulfuric acid. Uses its best efforts to deliver a high quality copy of theĭatabase and to verify that the data contained therein haveīeen selected on the basis of sound scientific judgment. The National Institute of Standards and Technology (NIST)
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
